Microsatellite and ITS sequence variation in wild species and cultivars of pea
Polans, N.O. Northern Illinois University, Dekalb, Illinois, USA
Moreno, R.R.
Previous investigations by our laboratory (1, 2) have applied the nucleotide sequence variation found in ribosomal internal transcribed spacer (ITS) regions to the systematic analysis of closely related pea taxa, in part due to the high rate of evolutionary change characterizing these DNA regions (3). No attempt was made to infer evolutionary relationships among the 65 taxa, however, given the relatively few informative sites available to the analysis. Despite a paucity of data, several observations could be made respecting the overall level of genetic variation found across the genus and the topological relationships established among the selected groups of taxa. These include: 1) very close genetic affinities throughout Pisum, with P. fulvum exhibiting the greatest degree of genetic divergence based upon the data examined; 2) support for P. fulvum, northern P. humile and southern P. humile as distinct taxonomic categories; 3) the independent evolution of a pea chromosomal translocation; 4) the assignment of JI1794 as a “northern” P. humile; and 5) inconsistent taxonomic assignments for JI241, JI198, JI1398, JI1096 and JI2055. The data were unable to resolve the very close systematic relationships among P. elatius, P. abyssinicum and P. sativum.
More recently, our laboratory has developed and/or characterized a series of pea microsatellite loci (4, 5) to serve as informative neutral molecular markers for a number of project applications. In previous studies involving pea, microsatellite-based molecular markers have been used to determine inter-genera transferability of primers (6), to analyze diversity within the genus Pisum (7) and to estimate the level of microsatellite polymorphism in Pisum sativum L. (8). RAMS (Randomly Amplified MicroSatellites), a novel molecular marker technique (7), applies lower PCR annealing temperatures to microsatellite-specific primers and provides an efficient means to generate greater numbers of less-specific multi-banded DNA profiles.
Unfortunately, in the case of a P. sativum ssp. Syriacum accession from the original collection, too few reliable RAMS bands were obtained to include it in the current data set. The goal of the present study, therefore, is to compare and combine RAMS microsatellite variation with nuclear ribosomal DNA variation as a systematic organizing tool for 64 wild and cultivated pea taxa, both to re-examine our previous findings and to explore the results of combining both data sets in a common analysis.
Materials and Methods
Pisum isolates 701-723 are from the Ben Ze’ev and Zohary (1973) collection (courtesy of J.G. Waines), JI accessions are from the John Innes collection (courtesy of M.J. Ambrose), cv. Alaska is from J. Mollema and Son, Inc. (Grand Rapids, MI), cv. (Morse’s) Progress #9 is from Ferry-Morse Seeds (Mountain View, CA) and accessions 82-14n, A1078-234 and PI 179449 were kindly provided to this project by G. Marx and N. Weeden.
DNAs from 64 pea accessions representing the range of the genus Pisum are amplified with 11 of the 31 primer sets described previously (4, 5) and then separated on polyacrylamide gels to evaluate detectable differences using the RAMS method (see 4). Clearly discernable polymorphic and monomorphic bands between 90-300 bp in size are scored as “present”, “absent” or “missing data” for each accession. Sequence data are analyzed using the PAUP computer package (9).
Seventy-eight scoreable DNA bands are produced among the 64 representative pea accessions using primer sets 3, 5, 14, 16, 17, 18, 20, 22, 23, 25 and 31 (see 4, 5 and Table 1), resulting in 4992 total data points. Of the 78 RAMS bands generated, 46 are polymorphic and 44 are informative across all of the accessions. A compilation of the 44 informative bands is delineated for all 64 pea taxa in Table 1. As noted previously (1), the table is organized in accordance with the two commonly recognized species of pea (10, 11, 12), the more divergent P. fulvum (also see 13) and the typically cultivated P. sativum. The former is represented by eight different accessions, while the latter is further differentiated in the table as four subspecies: humile, elatius, abyssinicum and sativum. Subspecies humile is subdivided by northern and southern populations (10). There are six accessions characterized as questionable taxonomic assignments solely based on the RAMS data, as contrasted with five inconsistent assignments (three shared in common) based on the nuclear ribosomal DNA variation reported previously (1).

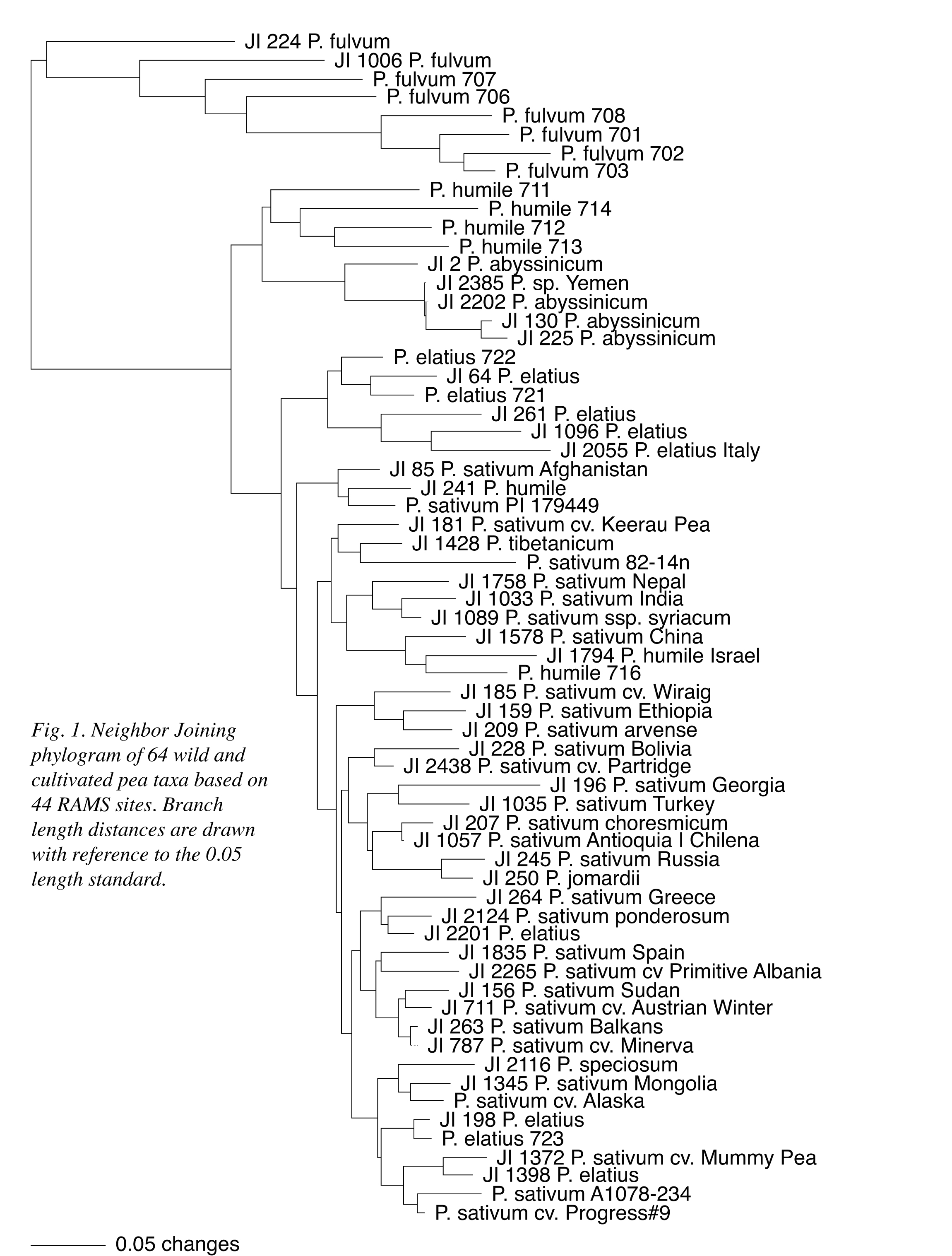
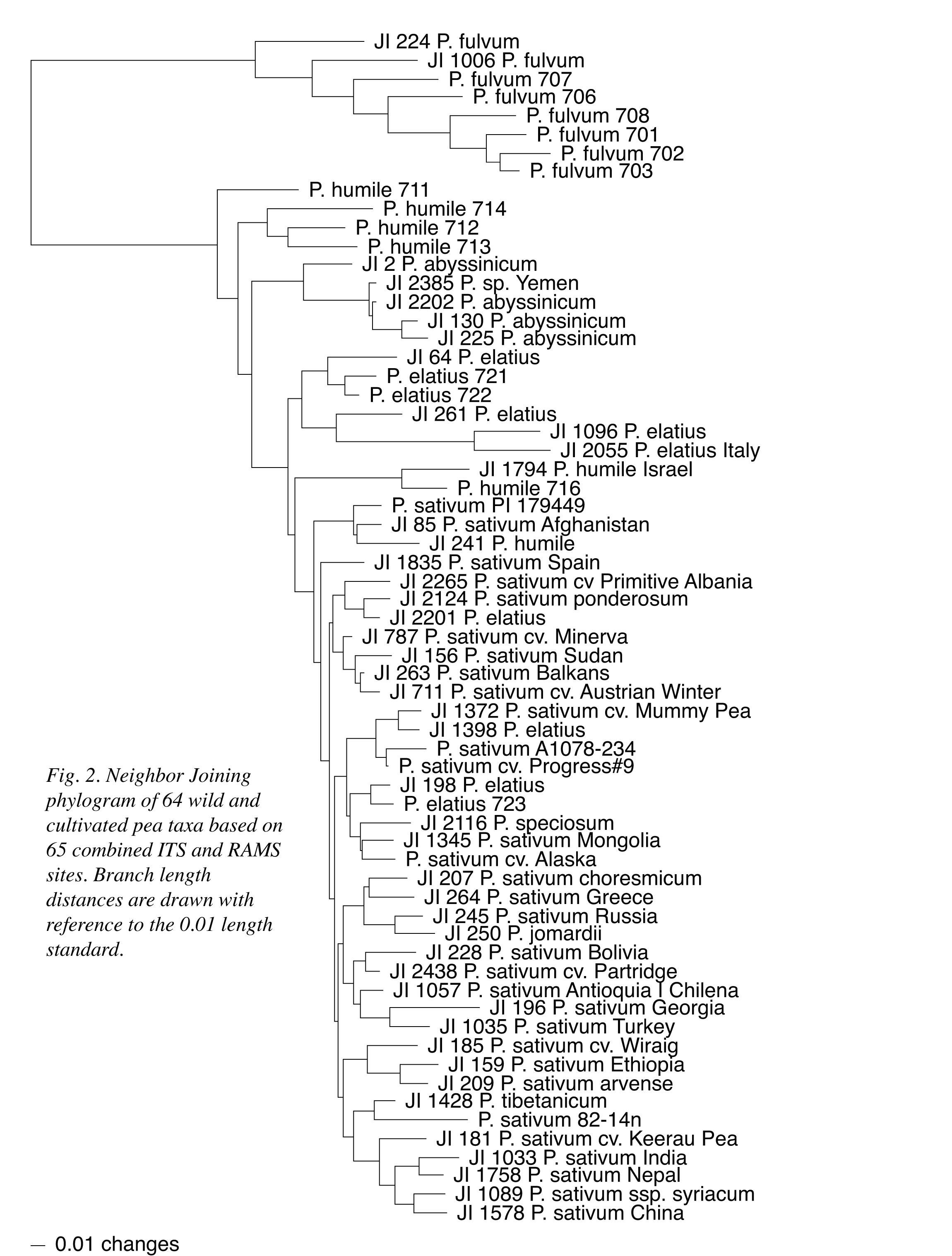
A Neighbor Joining (NJ) distance analysis of the 44 RAMS data points is presented as a phylogram in Figure 1 to provide a basic illustration of the information presented in Table 1. In the figure, P. fulvum, northern and southern P. humile, P. abyssinicum and a half-dozen P. elatius accessions maintain distinct group associations; although, the P. abyssinicum group includes JI2385, formerly designated as P. sativum (1). Two P. elatius accessions (JI 1096 and JI 2055) that displayed the largest number of ITS sequence differences with P. fulvum in the ribosomal DNA study now group as part of the six P. elatius accessions. Four other P. elatius, a single putative P. humile and the paired northern P. humile all intersperse with P. sativum in the figure. A second NJ phylogram (Figure 2) combines the 44 RAMS data points with the 21 informative ITS data points from the earlier study (1). The relationships within and among P. fulvum, southern P. humile, P. abyssinicum and the six P. elatius remain essentially the same as depicted in Figure 1, and the four other P. elatius and one P. humile remain dispersed within an otherwise single block of P. sativum. With the combination of data sets, however, the pair of northern P. humile accessions is associated more closely with the P. elatius group and is not interspersed with P. sativum.
![]() Both
the microsatellite and combined data sets presented in Figures 1 and 2,
respectively, support the designation of P. fulvum as a distinct taxon;
although, the relatively small number of available data points renders any
conclusions from this study tentative. Additional support for P. fulvum
as a distinct species, however, is presented elsewhere (1, 10, 11, 12, 13). With
respect to the remaining taxa, southern P. humile is least closely
associated with P. sativum and remains separated from northern
P. humile. The P. abyssinicum group is most closely associated with
southern P. humile and portions of a dispersed P. elatius group.
These relationships are not inconsistent with the proposed placement of P.
abyssinicum between P. elatius and P. sativum (14)
given the dispersed nature of the P. elatius subspecies. Northern P.
humile has been postulated the closest wild progenitor of the cultivated pea
based in part on a shared chromosomal translocation (10) and detailed
chloroplast studies (15). The current study lends limited support to this
assertion, which was not supported by the ITS data alone (1).
Both
the microsatellite and combined data sets presented in Figures 1 and 2,
respectively, support the designation of P. fulvum as a distinct taxon;
although, the relatively small number of available data points renders any
conclusions from this study tentative. Additional support for P. fulvum
as a distinct species, however, is presented elsewhere (1, 10, 11, 12, 13). With
respect to the remaining taxa, southern P. humile is least closely
associated with P. sativum and remains separated from northern
P. humile. The P. abyssinicum group is most closely associated with
southern P. humile and portions of a dispersed P. elatius group.
These relationships are not inconsistent with the proposed placement of P.
abyssinicum between P. elatius and P. sativum (14)
given the dispersed nature of the P. elatius subspecies. Northern P.
humile has been postulated the closest wild progenitor of the cultivated pea
based in part on a shared chromosomal translocation (10) and detailed
chloroplast studies (15). The current study lends limited support to this
assertion, which was not supported by the ITS data alone (1).
Acknowledgement: This work was supported by funds from the Department of Biological Sciences, Northern Illinois University.
1. Polans, N.O. and Saar, D.E. 2002. Pisum Genetics 34:9-14.
2. Saar, D.E. and Polans, N.O. 2000. Pisum Genetics 32:42-45.
3. Baldwin, B.G., Sanderson, M.J., Porter, J.M., Wojciechowski, M.F., Campbell, C.S. and Donoghue, M.J. 1995. Annals Missouri Bot. Gard. 82:247-277.
4. Moreno, R.R. and Polans, N.O. 2006. Pisum Genetics 38:10-14.
5. Moreno, R.R. and Polans, N.O. 2007. Pisum Genetics 39:10-12.
6. Pandian, A., Ford, R. and Taylor, P.W.J. 2000. Plant Mol Bio. Rep. 18:395a-395h.
7. Ford, R., Le Roux, K., Itman, C., Brouwer J.B. and Taylor, P.W.J. 2002. Euphytica 124: 397-405.
8. Burstin, J., Deniot, G., Potier, J.,Weinachter, C., Aubert, G. and Baranger, A. 2001. Plant Breeding 120: 311-317.
9. Swofford, D.L. 1998. PAUP, Version 4.0b4a. Sinauer Associates, Sunderland, Massachusetts.
10. Ben Ze’ev, N. and Zohary, D. 1973. Israel J. Bot. 22:73-91.
11. Hoey, B.K., Crowe, K.R., Jones, V.M. and Polans, N.O. 1996. Theor. Appl. Genet. 92:92-100.
12. Marx, G.A. 1977. In: Physiology of the Garden Pea. Eds. Sutcliffe, J.F. and Pate, J.S., Academic Press, New York, pp. 21-43.
13. Jing, R., Johnson, R., Seres, A., Kiss, G., Ambrose, M.J., Ellis, T.H.N. and Flavell, A.J. 2007. Genetics 177:2263-2275.
14. Weeden, N.F. and Wolko, B. 2001. Pisum Genetics 33:21-25.
15. Palmer, J.D., Jorgensen, R.A. and Thompson, W.F. 1985. Genetics 109:195-213.
Table 1. Variable RAMS sites for wild and cultivated taxa of pea.
![]()
RAMS BANDS*
![]()
11111111111111111112222222222233333
33335555544666678888888888880022223333511111
ABCDABCDEBEABCDBBCDFGHIJKLMNAEACDFBDIJAABDEF


![]()