PISUM Genetics
2003—Volume 35
Research Papers
Stipule morphologies of the sinuate leaf (sil) mutants
Husbands, A., Emirzade, T. and
DeMason, D.
DeMason, D.
Botany and Plant Sci., Univ. of California
Riverside, CA 92521, USA
Riverside, CA 92521, USA
Form and development of the
pinnately compound leaf of pea are
genetically controlled by a number
of known genes (6). Some genes
appear to affect only blade
development and others to affect
only stipule development. The best
known blade mutations are afila (af),
which transforms the proximal
leaflets into branched tendrils; and,
tendrilless (tl), which transforms the
distal tendrils into leaflets (4). A
mutation that affects both blade and
stipule, known as sinuate leaf (sil),
was originally recognized in an af
pinnately compound leaf of pea are
genetically controlled by a number
of known genes (6). Some genes
appear to affect only blade
development and others to affect
only stipule development. The best
known blade mutations are afila (af),
which transforms the proximal
leaflets into branched tendrils; and,
tendrilless (tl), which transforms the
distal tendrils into leaflets (4). A
mutation that affects both blade and
stipule, known as sinuate leaf (sil),
was originally recognized in an af
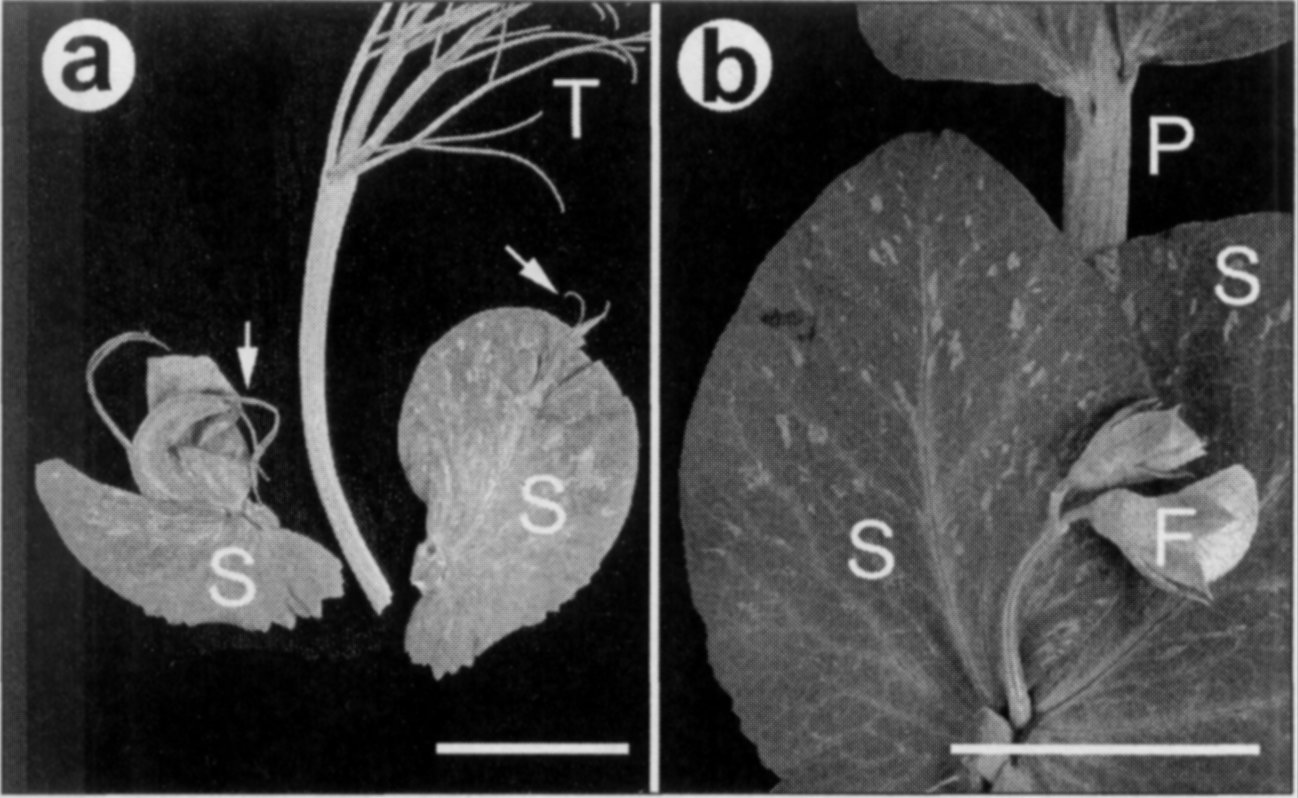
line. af/af sil/sil plants possess
"adventitious tendrils arising from a
cleft in the distal tip of stipules"
(Fig. la; 5). Marx (5) originally
"adventitious tendrils arising from a
cleft in the distal tip of stipules"
(Fig. la; 5). Marx (5) originally
Fig. 1. sil stipule phenotypes. a. stipule from lower node showing
adventitious tendrils forming from cleft at stipule tip as originally described
by Marx (5); b. stipules at flowering node showing no phenotype. Bars =
2.5 cm. B — leaf blade: F — flower buds: P — Petiole: S — stipule.
adventitious tendrils forming from cleft at stipule tip as originally described
by Marx (5); b. stipules at flowering node showing no phenotype. Bars =
2.5 cm. B — leaf blade: F — flower buds: P — Petiole: S — stipule.
described the phenotypes of sil
mutants in Af/Af, Af/af and af/af backgrounds. He reported that Af/- sil plants had sinuate or undulated
leaflet and stipule margins but that only af/af sil plants had stipule tip modifications. He concluded that there
were clear gene interactions between Af and Sil, but that anatomical or histological studies were necessary to
develop a better understanding of them. In an attempt to address Marx's challenge, we recreated these three
lines and started making observations of stipule form changes during shoot ontogeny in preparation for
selecting material for anatomical preparations. Here we correct Marx's observations on stipular morphologies
present on Af/Af sil/sil and Af/af sil/sil plants and provide a hypothesis to explain the interactions between Af
and Sil
leaflet and stipule margins but that only af/af sil plants had stipule tip modifications. He concluded that there
were clear gene interactions between Af and Sil, but that anatomical or histological studies were necessary to
develop a better understanding of them. In an attempt to address Marx's challenge, we recreated these three
lines and started making observations of stipule form changes during shoot ontogeny in preparation for
selecting material for anatomical preparations. Here we correct Marx's observations on stipular morphologies
present on Af/Af sil/sil and Af/af sil/sil plants and provide a hypothesis to explain the interactions between Af
and Sil
Materials and Methods
Two sil lines, W6 15137 (A Af b er f fs I pal sil) and W6 15110 (a af i sil) were obtained from the Marx
collection at the USDA Western Regional Plant Introduction Station, Pullman, Washington and were the
parental lines in this study. W6 15110 was used as the female and W6 15137 was used as the male parent.
The F1 plants were grown and allowed to self. F3 seedlings from individual F2 plants were phenotyped to
identity the genotype of the parental line. Seed was separated into three genotypic categories: Af/Af sil/sil,
af/af sil/sil, and Af/- sil/sil. For observations and photography, plants were grown in a standard greenhouse in
the fall (short days) when the stipular phenotypes were most dramatic. F4 seeds of each genotype were
planted during the first week of October and developed under natural lighting conditions. Greenhouse
temperatures were approximately 20° in the day and 15° at night.
collection at the USDA Western Regional Plant Introduction Station, Pullman, Washington and were the
parental lines in this study. W6 15110 was used as the female and W6 15137 was used as the male parent.
The F1 plants were grown and allowed to self. F3 seedlings from individual F2 plants were phenotyped to
identity the genotype of the parental line. Seed was separated into three genotypic categories: Af/Af sil/sil,
af/af sil/sil, and Af/- sil/sil. For observations and photography, plants were grown in a standard greenhouse in
the fall (short days) when the stipular phenotypes were most dramatic. F4 seeds of each genotype were
planted during the first week of October and developed under natural lighting conditions. Greenhouse
temperatures were approximately 20° in the day and 15° at night.
Results
Distinct morphological features were characteristic of stipule tips on plants of each of the genotypes
generated in this study. There was a progressive change in expression from node to node, up the plant. On
adult flowering plants, the stipules at lower, or juvenile, nodes displayed nearly normal phenotypes, which
generated in this study. There was a progressive change in expression from node to node, up the plant. On
adult flowering plants, the stipules at lower, or juvenile, nodes displayed nearly normal phenotypes, which
