Strategies for improving the iron nutritional quality of seed crops: lessons learned from the study of unique iron-hyperaccumulating pea mutants
Michael A. Grusak USDA/ARS Children’s Nutrition Research Center, Dept. of Pediatrics
Baylor College of Medicine, 1100 Bates Street, Houston, TX, 77030
There is growing interest to improve the nutritional quality of our food supply, especially with respect to essential micronutrient minerals, such as iron (Fe) (1, 10, 11). Recent estimates indicate that one-third of the world’s population is at risk for iron-deficiency induced anemia, a condition considered to be the most prevalent nutrient-related human disease on the planet (27). The primary cause of this problem is that most individuals, especially in developing countries, subsist on diets comprised mainly of plant foods; unfortunately, these diets are not supplying adequate daily intakes of Fe. Whereas plants can provide almost all the essential nutrients required by humans (10), plant Fe concentration is generally low relative to that of most animal-derived foods (Table 1), and the form of Fe in plants is poorly absorbed in the gut (24).
Table 1. Iron concentrations for plant and animal food sources. Concentrations were derived from ref. 23 and represent values for a range of common foods from each food group. Note that edible portions are reported, such that water content will vary among reported items.
|
Food group |
mg Fe/100 g edible portion |
|
Cereal grains |
0.4 – 2.1 |
|
Legume seeds |
1.5 – 2.9 |
|
Pea seeds |
1.5 |
|
Leafy vegetables |
0.7 – 3.6 |
|
Root crops |
0.2 – 0.8 |
|
Fruit |
0.1 – 0.3 |
|
Red meat |
2.7 – 10.7 |
|
|
|
Although increased consumption of animal products would help to alleviate iron deficiency in at-risk populations, there are numerous religious, cultural, and/or economic factors that make this an unrealistic possibility. Instead, we and others have suggested that enhancing the Fe density of commonly consumed plant foods would be a sound, sustainable approach to improve human iron status (1, 7, 11). Furthermore, because seeds are the foundation of most plant-derived food products, recent efforts have focused on seed crops, with researchers interested in identifying existing germplasm, or in developing new cultivars, with enhanced Fe concentration.
In order to facilitate cultivar development, our research has focused on determining the mechanisms and regulatory processes responsible for Fe acquisition, partitioning, and eventual transport to developing seeds. We believe this understanding is crucial to be able to effectively manipulate the plant, and to override existing homeostatic processes that regulate plant Fe content. We have directed our efforts towards pea, because of the availability of two Fe-hyperaccumulating pea mutants, brz and dgl (6, 18) and because of the promise they offered in helping to understand various aspects of whole-plant iron nutritional physiology. The study of these mutants has taught us much about how Fe content could potentially be altered in the pea crop itself, whose seeds have a low Fe concentration relative to that of other legumes (Table 1); additionally, the mutants are serving as useful models for the development of Fe improvement strategies in other important legumes, and even in various cereal crops.
Whole-plant iron homeostasis
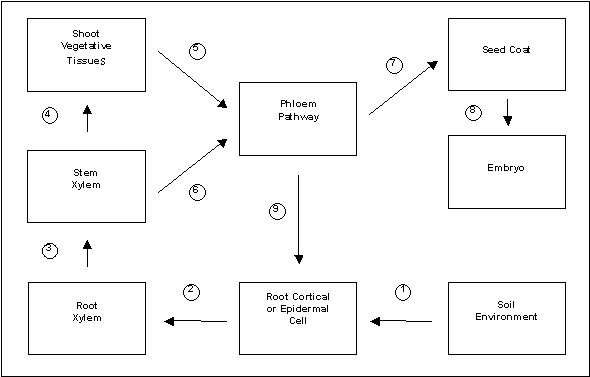
In pea, as in other plants, specialized mechanisms have evolved to control the acquisition and handling of Fe at the cellular, tissue, and whole-plant level (Fig. 1). These mechanisms constitute the homeostatic processes that assist the plant in attempting to obtain adequate Fe nutrition, while at the same time preventing excess Fe accumulation and/or toxic damage (13). Because Fe is an essential mineral, proper homeostatic regulation of Fe is critical to the general growth and reproductive success of the individual; it also contributes significantly to the eventual Fe content of edible tissues.

Fig. 1. Scheme of whole-plant Fe homeostasis in pea. Regulatory processes that would influence the movement of Fe from one compartment to the next include: (1) Fe(III)-chelate reductase and Fe2+ transporter; (2) intracellular transport and Fe metabolism; (3) cell-wall-cationic binding sites; (4) transpiration rates of vegetative tissues; (5) intracellular Fe metabolism/sequestration and phloem loading capacity, including the expression of an Fe-chelator; (6) xylem-to-phloem exchange; (7) phloem transport capacity of photoassimilates from a given source region; (8) seed coat metabolism and Fe trafficking to the embryo apoplast; (9) shoot-to-root communication of shoot Fe status.
For the brz and dgl pea mutants, it appeared that each might contain a defect in some critical Fe homeostasis component because both demonstrate uncontrolled Fe acquisition when provided with an available source of Fe. Leaf Fe levels in wild type pea usually range from 100 to 150 mg/g DW, while Fe concentrations can reach 5,000 to 10,000 mg/g DW in mutant leaves (18, 25). These high levels of Fe lead to necrotic lesions in leaves (17) that reduce photosynthetic capacity and eventually result in plant death. (An image of Fe-toxic brz leaves can be seen at: http://www.ars-grin.gov/ars/PacWest/Pullman/GenStock/ pea/brz.html). Thus, the mutants are unable to survive and successfully reproduce in most soil types, although they have been shown to have an adaptive advantage in soils with low Fe availability (2, 6). Segregation analysis demonstrates that the two mutations occur at different alleles and that they function in a semi-dominant manner; F1 hybrids exhibit intermediate leaf Fe concentrations (18).
Fe is acquired at the root-soil interface. Thus, early efforts with the mutants focused on their root physiology and the possibility that one or several root processes might be altered. It previously had been established that root Fe acquisition in dicots, such as pea, involved the obligate reduction of ferric iron (Fe3+; the predominant form in most oxygenated soils) to ferrous iron (Fe2+) through the action of a plasmalemma-localized reductase (3). This enzyme provides the Fe2+ substrate for an Fe2+ transport protein (4), also located on the plasmalemma. Previous work with various species had shown that when plants are challenged with Fe-deficiency stress, the activity of the root Fe(III) reductase is enhanced (21, 22). Interestingly, root Fe(III) reductase activity in the brz and dgl pea mutants is elevated, relative to Fe-nourished wild type plants, even when the mutants are Fe-replete (14, 15). In general, the mutants exhibit several activated root processes (when grown with or without Fe) that are characteristic of Fe-deficient wild type pea (elevated reductase activity, elevated Fe2+ transport capacity, enhanced rhizosphere acidification) (5, 12, 14, 15, 26), and thus brz and dgl have been described as functional Fe-deficiency mutants.
Regulation of root iron acquisition
The constitutively elevated root processes (exhibited by the mutants) provided a means to assess the relative importance of Fe reduction versus Fe influx in the overall micronutrient nutrition of the plant. Studies with the brz mutant and wild type pea (cv. Sparkle) demonstrated that the root Fe(III) reductase was the rate-limiting physiological process with respect to Fe acquisition in the roots (16). Kinetic analysis of Fe2+ influx showed that transport capacity would saturate only at Fe2+ concentrations far higher than those achievable by the Fe(III) reductase under normal soil conditions. Thus, the root system’s ability to reduce soluble Fe(III)-compounds was critical in regulating the availability of Fe2+ to the transporter, and total Fe to the plant.
These results were important, because they helped direct our attention towards understanding how reductase activity was regulated by the whole organism. With respect to the brz and dgl mutants, constantly elevated root reductase activity explained why the plants could hyperaccumulate Fe. These observations also suggested that reductase activity was normally low in most Fe-replete plants, and that it was induced towards higher activity and/or higher expression when plant Fe requirements were not being met. In other words, regulation involved turning on an otherwise low reductase capacity, rather than turning off a high capacity.
This type of regulation makes sense when the particular nutrient, if absorbed in excess, can lead to toxic damage of plant tissues. But how does the bulk of the organism which requires Fe (i.e., the vegetative and reproductive tissues of the shoot) communicate its Fe needs to those tissues that are responsible for Fe absorption (i.e., the roots)? Grafting studies with the mutants helped address this question, as reciprocal shoot-root grafts revealed that the elevated root reductase activities observed in Fe-replete plants were controlled by some property associated with the mutant shoots (14). For grafted plants maintained on Fe, mutant shoots could promote high reductase activities in wild type root systems, but activities were low in graft combinations bearing wild type shoots on mutant root stocks. Thus, both the brz and dgl mutations were apparently inducing some change in shoot tissues that resulted in the transmission of a reductase-inducing signal to the roots. Additional studies suggested that this signal was promotive in nature, and that it was transmitted via the phloem pathway (14). Furthermore, experiments using wild type pea also substantiated the involvement of a phloem-mobile signal in the regulation of root Fe-acquisition processes (9), and helped promote the concept that shoot-to-root communication, with respect to Fe homeostasis, is a general phenomenon in all plants (13, 22).
The nature of this signal has yet to be determined, although it is clear that the mutants can serve as useful tools in the identification of this compound. Recent and ongoing efforts have focused on the development of mass spectrometry and other analytical techniques to characterize chemical constituents of the phloem sap, in both wild type and mutant peas (19).
Phloem loading and transport to seeds
Whereas root absorption of Fe is clearly important for general nutrition, how the plant handles, partitions, and transports Fe throughout the whole organism (Fig. 1) has significance to the Fe content of edible organs. With respect to the pea seed, although both xylem and phloem elements are contiguous between the vegetative plant and the seed coat, only the phloem pathway is involved in the movement of nutrients to developing seeds. This is because the confining nature of the pod walls results in a seed microenvironment in which the relative humidity is maintained at or near 100%. Thus, there is no driving force for transpiration from the surface of the seeds, and no xylem flow into the seed coats.
With phloem transport responsible for seed Fe import, we again utilized the mutants to address the question of how Fe was loaded into the phloem pathway, and how this process might be regulated. Initially, we demonstrated that leaves, stipules, and pod walls, all had the capacity to load Fe and effect its phloem transport to developing seeds (8). We also showed that Fe was transported to seeds throughout the whole period of seed growth (8). However, the amount of Fe mobilized to seeds was found to be tightly regulated. Studies with the brz mutant revealed that with even a 36-fold increase in leaf Fe content, relative to wild type, no increase was measured in the Fe concentration of seeds (8). Both brz and its wild type parent (cv. Sparkle), when grown with sufficient or excess Fe, exhibit seed Fe concentrations of 70 to 80 mg/g DW (this is roughly equivalent to the 1.5 mg/g edible in Table 1). These results, along with other theoretical considerations, led to the hypothesis that Fe is phloem loaded in a chelated form, and that the expression/synthesis of the endogenous chelator is an important factor in the control of Fe transport to seeds.
For the dgl mutant, a different seed outcome was observed. Providing excess Fe to the plants resulted in seed Fe concentrations as high as 250 mg/g DW (20), or roughly 3.5-fold that of wild type pea seeds. Based on our previous results, this outcome could be explained by the dgl mutation conditioning for the overexpression of an endogenous Fe-chelator, or perhaps the expression of a novel chelator with the ability to load Fe into the phloem pathway. As with the phloem-mobile signal compound, we have yet to identify the endogenous and/or novel Fe-chelators involved in phloem transport, but their identification is an active focus of our current research program.
Strategies for improving the iron nutritional quality of seeds
The mutant results are very promising, as they demonstrate that at least in pea, the endogenous Fe homeostatic machinery can be manipulated to enhance vegetative Fe concentration (as shown with brz or dgl), as well as seed Fe concentration (as shown with dgl). However, we suspect that Fe homeostasis could be manipulated to a similar extent in other crops to enhance their Fe content as well.
To do this for seeds, it appears that the expression of a phloem-mobile Fe chelator must be upregulated in order to mobilize more Fe from vegetative tissues. Interestingly, in the case of wild type pea, we have shown that 75% of the total shoot Fe already is transported to the seed fraction, such that even if all the remaining Fe could be mobilized (which is unlikely), the best case outcome would be an increase in seed Fe concentration of 33%. Clearly, more Fe would need to be absorbed into the plant in order to serve as co-substrate for phloem loading.
With respect to the dgl mutant, the increased whole-plant uptake of Fe explains why this plant can demonstrate a 3.5-fold increase in seed Fe concentration. Unfortunately, the unregulated root Fe uptake in dgl also leads to toxic stress in most tissues. Thus, for the improvement of pea, and probably for most other legumes, a targeted manipulation of a phloem-mobile chelator would need to be coordinated with an upregulation of root Fe acquisition (e.g., via the signal molecule). Ideally, if these manipulations could be synchronized with reproductive development, any excess Fe entering the plant could then be transported immediately to the seeds. However, it should be noted that additional changes may need to be effected in seeds themselves, as not all the excess Fe in dgl seeds has been found safely sequestered in the Fe storage protein, ferritin (20). This unsequestered Fe, and its potential for free radical generation, may explain why seed viability declines rapidly in dgl seeds (<10% germination within 3 years, relative to >95% in wild type; Grusak, unpublished results).
With respect to the improvement of cereal grains, we believe that the results of our mutant investigations also have application. We presume that a similar degree of Fe transport regulation occurs at the point of phloem loading in grasses, especially since the Fe concentration of most cereal grains tends to be lower than that of the legumes (Table 1). Whether the nature of the phloem-mobile Fe chelator is identical in all plants remains to be determined, but one can speculate that the availability of an Fe chelator would need to be increased in source leaves of cereal crops, in order to enhance seed Fe content. Interestingly, we have found that the amount of Fe partitioned to grains in the mature rice plant is only 4% of total shoot Fe (Grusak, unpublished results); therefore, it appears that the manipulation of phloem Fe loading would not have to be linked to increased whole-plant Fe uptake in this plant. There appears to be an ample reservoir of excess Fe already present for phloem loading, assuming this reservoir is in an available and accessible form.
In summary, the analysis of unique, Fe-hyperaccumulating pea mutants has contributed significantly to our understanding of Fe nutritional physiology. Studies with the mutants have taught us that whole-plant Fe homeostasis involves a number of complex phenomena (Fig. 1), and that the controlled manipulation of Fe content at the whole-plant level will require modifications at several points along the plant continuum. We anticipate that further progress will be made with the brz and dgl mutants in the coming years, but we also are hopeful that other pea mutants will be identified that might reveal additional pieces of the whole-plant Fe puzzle.
Acknowledgements: The results discussed in this review were supported in part by the U.S. Department of Agriculture, Agricultural Research Service (CRIS No. 6250-21520-002-00D and CRIS No. 6250-21520-041-004), the U.S. Department of Agriculture, Cooperative State Research, Education, and Extension Service, National Research Initiative (Award No. 94-37100-0832), and the National Science Foundation (Award No. IBN-9630341). The contents of this publication do not necessarily reflect the views or policies of the US Department of Agriculture, nor does mention of trade names, commercial products, or organizations imply endorsement by the US Government.
1. Bouis, H. 1996. Nutr. Rev. 54: 131-137.
2. Cary, E.E., Norvell, W.A., Grunes, D.L., Welch, R.M. and Reid, W.S. 1994. Agron. J. 86: 938-941.
3. Chaney, R.L., Brown, J.C. and Tiffin, L.O. 1972. Plant Physiol. 50:298-213.
4. Cohen, C.K., Fox, T.C., Garvin, D.F. and Kochian, L.V. 1998. Plant Physiol. 116: 1063-1072.
5. Fox, T.C., Shaff, J.E., Grusak, M.A., Norvell, W.A., Chen, Y., Chaney, R.L. and Kochian, L.V. 1996. Plant Physiol. 111: 93-100.
6. Gottschalk, W.G. 1987. Pisum Newsletter 19: 9-11.
7. Graham, R. D. and Welch, R. M. 1996. Breeding for staple food crops with high micronutrient density. International Food Policy Research Institute, Washington, D.C., 79 pp.
8. Grusak, M.A. 1994. Plant Physiol. 104: 649-655.
9. Grusak, M.A. 1995. Planta 197: 111-117.
10. Grusak, M.A. and DellaPenna, D. 1999. Ann. Rev. Plant Physiol. Plant Mol. Biol. 50: 133-161.
11. Grusak, M.A., DellaPenna, D. and Welch, R.M. 1999. Nutr. Rev. 57: S27-S33.
12. Grusak, M.A., Kochian, L.V. and Welch, R.M. 1993. Amer. J. Bot. 80: 300-308.
13. Grusak, M.A., Marentes, E. and Pearson, J.N. 1997. Field Crops Res. 60: 41-56.
14. Grusak, M.A. and Pezeshgi, S. 1996. Plant Physiol. 110: 329-334.
15. Grusak, M.A., Welch, R.M. and Kochian, L.V. 1990. Plant Physiol. 93: 976-981.
16. Grusak, M.A., Welch, R.M. and Kochian, L.V. 1990. Plant Physiol. 94: 1353-1357.
17. Guinel, F.C. and LaRue, T.A. 1990. Can. J. Bot. 68: 1340-1348.
18. Kneen, B.E., LaRue, T.A., Welch, R.M. and Weeden, N.F. 1990. Plant Physiol. 93: 717-722.
19. Marentes, E. and Grusak, M.A. 1998. J. Exp. Bot. 49: 903-911.
20. Marentes, E. and Grusak, M.A. 1998. Seed Sci. Res. 8: 367-375.
21. Marschner, H. and Römheld, V. 1994. Plant Soil 165: 261-274.
22. Schmidt, W. 1999. New Phytol. 141: 1-26.
23. USDA, ARS. 1999. Nutrient Database for Standard Reference, Release 13. http://www.nal.usda.gov/ fnic/foodcomp
24. van den Heuvel, E.G.H.M., van Dokkum, W. and Schaafsma, G. 1997. Food Rev. Int. 13: 91-102.
25. Welch, R.M. and LaRue, T.A. 1990. Plant Physiol. 93: 723-729.
26. Welch, R.M., Norvell, W.A., Schaefer, S.C., Shaff, J.E. and Kochian, L.V. (1993) Planta 190: 555-561.
27. Yip, R. 1994. J. Nutr. 124: 1479S-1490S.