| Gorel, F.L., Kosterin, O.E. and Berdnikov, V.A |
Institute of Cytology and Genetics Novosibirsk 630090 Russia |
Structural heterozygotes for translocations are of interest for gene mapping in several respects. First, the translocation breakpoints (T-points) behave as genetic markers manifested by pollen semisterility in heterozygotes versus pollen fertility in both types of structural homozygotes. Second, two intact and two interchange chromosomes are involved in the analysis. Third, translocations suppress recombination in the vicinity of T-points, facilitating recognition of linkage with distant markers. Finally, tertiary trisomics can be found among the progeny of translocation heterozygotes from selfing, permitting trisomy analysis.
We have performed a large experiment with the goal of adding markers to the genetic map in the regions adjacent to the centromeres of linkage groups II and V. For the sake of clarity in this manuscript we will use chromosome numbers paralleling the linkage group numbers as published in the most recent version of the pea genetic map (4). It is well known (1-3) that the centromeric regions are in the vicinity of T-points of Hammarlund translocation, involving chromosomes 2 and 5. We decided to treat the heterozygotes for this translocation by EMS and then identify new mutations linked to T-points and to the markers situated in the centromeric regions of chromosomes 2 and 5 of the normal karyotype. The plant material used was hybrid stock designated ‘Tau-2.’ This hybrid was generated from the line WL1476 [translocated karyotype (K)] with genotype Cri, Gp, A, His(2-6)
2121 (1), and a partially inbred line with normal karyotype (N) and genotype cri, gp, a, His(2-6)1123. The latter stock had a complicated pedigree, being in general derived from the lines WL1238, WL1018, our line SG, and a mutation cri induced in our laboratory with EMS in line SG.In the hybrid Tau-2 (karyotype N/K) a cross structure is resolved in meiosis I, shown on Fig. 1. The hyperploid gametes carry the additional minor interchange chromosome 5
2, composed of the short arms of the chromosomes 2 and 5. After combining with euploid gametes, viable and fertile trisomics are formed (1-3).
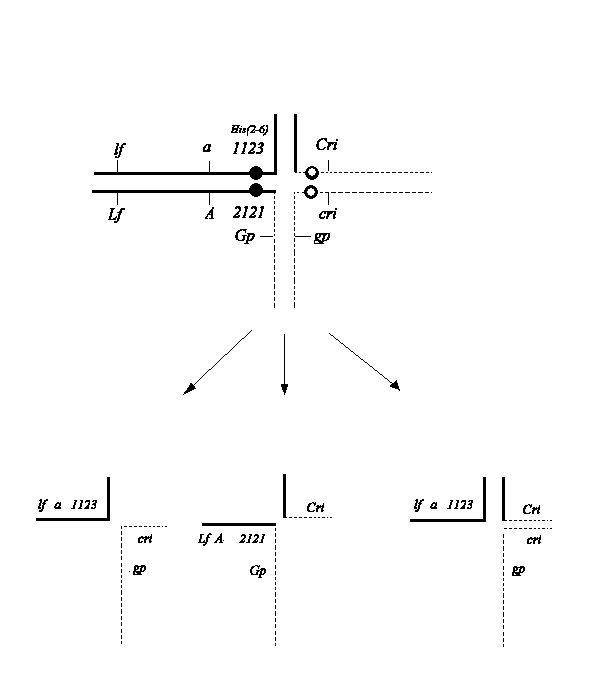
Fig. 1. A scheme of a translocation cross forming in meiosis in the hybrid Tau-2 and the types of gametes produced. It should be noted that only one class (N+52) of hyperploid gametes is produced by the structural heterozygote (3).
Selfing Tau-2 results in five main classes of progeny: structural homozygotes N/N for the normal and interchange (K/K) karyotype, structural heterozygotes (N/K), and tertiary trisomics with karyotypes N/N+52 and N/K + 52. The recessive alleles cri, gp, and a are located near the T-point of chromosomes of normal karyotype, while the dominant alleles reside on the interchange chromosomes. Therefore we can easily distinguish structural homozygotes N/N (with the phenotype cri gp a) and trisomics N/N+52 (phenotype Cri gp a). Trisomics (Cri gp a) can be confused with the very rare cross-overs between the breakpoint and cri, but they can be easily recognized by slightly reduced pollen fertility, large bracts (3), and by examination of their progeny. Class N/K (with the phenotype Cri Gp A) can be distinguished from K/K by pollen semisterility and codominant expression of histone H1 subtypes (1123+2121) encoded by the gene cluster His(2-6). This same phenotype can be exhibited by another class of trisomics N/K+52. However, this last genotype can be recognized by a weak growth habit and large bracts.Among 800 plants produced from selfing the structural heterozygote, Tau-2, we obtained 44 trisomics (N/N+5
2). In addition we identified seven cross-over events between the T-point and gp, three between the T-point and a, none between the T-point and cri or between T-point and His(2-6).We imbibed 2165 F
4 seeds from Tau-2 in 0.15% solution of EMS overnight and grew the resulting plants hydroponically in the greenhouse. Among the M1 plants only structural heterozygotes were selected and their progeny germinated. Similarly, among M2 plants only structural heterozygotes (one M2 plant of each family) were allowed to produce seeds. By this procedure we eliminated about half of the mutations not linked to the T-points. The analysis of new mutations was carried out on M3 families grown in the field. We paid particular attention to families in which visible alterations of phenotype co-segregated or were negatively correlated with markers cri and a. Several mutations in the vicinity of the T-points were obtained. Here we report the results for one of them, a chlorophyll mutant of the xantha type.Among the M
3 families we found one in which 9 of 10 plants with phenotype cri had no chlorophyll but, judging by their yellow color, normal level of carotinoids. Another 34 plants of this family were normal green (Table 1). This new chlorophyll mutation was called golden (gld). All the nine gld plants died after producing 5 to 6 nodes but no flowers; however, absence of an anthocyanin ring at the base of the stipules indicated the phenotype a. In addition, all the mutants had a His(2-6) haplotype 1123. Thus, all mutants clearly had a normal karyotype (N/N). The plants with phenotype Cri Gld exhibited three phenotypic classes with respect to other markers (Table 1). Three, presumably trisomics, had white flowers with large bracts and the haplotype 1123. Two other classes had colored flowers with vexillary or no bracts. The heterozygotes (1123/2121) had a semisterile pollen (karyotype N/K), while the homozygotes (2121/2121) had normal pollen. Table 1. Segregation for phenotype classes in the original M3 family (resulted from selfing of a structural heterozygote) in which the mutation gld was found.| 1123, a1 | 1123+2121, A | 2121, A | |
| cri, gld | 9 | 0 | 0 |
| cri, Gld | 1 | 0 | 0 |
| Cri, Gld | 3.2 | 17 | 13 |
1
- phenotype a was determined by absence of the anthocyanin ring.One can make two inferences from these results. The mutation is tightly linked to the markers on chromosomes 2 and 5 of the normal karyotype, and it resides in the short arm of either of the chromosomes 2 or 5, because addition of the small interchange chromosome reconstitutes the normal phenotype.
If the karyotype of the plants with phenotype Cri Gld a 1123 was indeed N/N+5
2 then their genotype with respect to the locus Golden should be gld/gld/Gld and their progeny should comprise diploids gld/gld and trisomics like the parents. Two presumed trisomic plants differed from their diploid counterparts by less vigor and fertility. Only one of them gave 20 seeds, that were sowed into a greenhouse bed and gave rise to 19 seedlings, 15 of which had the phenotype cri gld and four had the parental phenotype Cri Gld and flowers with large bracts. Cytological analysis of PMC of the four plants revealed a small additional chromosome.Unfortunately, the golden mutation appeared in a family in which the allele Gp was fixed in a homozygous state most probably due to a cross-over event between the T-point and the locus Gp happened in the M
2 stage. For restoration of the marker lost we crossed one trisomic plant with our original line HT1, a structural homozygote for the Hammarlund translocation with the long interchange chromosome marked with A, His(2-6)2121, lfa, and gp. This line was derived from the hybrid Tau-2 by fixation of two cross-over events between T-points and the markers mentioned. An F1 plant obtained had a phenotype lf A 2121+1123 Gp Cri.Among the F
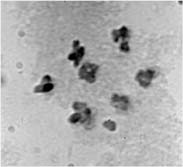
2 plants one can distinguish all karyotypes by their phenotypes: cri Gp 1123 a - for N/N, Cri gp 2121 A - for K/K, Cri Gp 2121+1123 A - for N/K, and Cri Gp 1123 a - for trisomics NN+52. Pollen fertility was an additional indication of karyotype: in structural homozygotes it was 100% fertile, in structural heterozygotes - 50% fertile, in trisomics - 80-90% fertile. The segregation of these phenotypes in F2 is shown in Table 2. We can see that all gld plants displayed markers of the normal karyotype. Pollen mother cells were analyzed from 10 of the 12 presumed trisomic plants and the presence of a small additional chromosome was confirmed (Fig. 2). The F3 progenies of the trisomics contained Cri Gld plants in an overall fraction of 0.26 (Table 3). Table 2. Segregation for phenotype classes in the F2 progeny of the cross of the trisomic Gld/gld/gld with the interchange line HT1.2121 A |
2121+1123 A |
1123a |
|
| cri gld Gp | 0 | 0 | 26 |
| cri Gld Gp | 0 | 0 | 1 |
| Cri Gld Gp | 0 | 83 | 12.1 |
| Cri Gld gp | 35 | 0 | 0 |

Fig.2. Metaphase I in PMC of trisomic plants among F
2 of the cross (trisomic Gld/gld/gld x HP1) Table 3. Segregation of phenotypes in the progeny of trisomics from F2 explained in Table 2No of trisomic |
cri gld |
cri Gld |
Cri Gld |
0 1 |
22 |
0 |
0 3 |
0 2 |
23 |
0 |
0 6 |
0 3 |
36 |
0 |
16 |
0 4 |
11 |
1 |
0 2 |
0 5 |
31 |
1 |
18 |
0 6 |
45 |
1 |
18 |
0 7 |
14 |
0 |
0 5 |
0 8 |
25 |
0 |
0 9 |
0 9 |
19 |
0 |
0 5 |
10 |
17 |
0 |
0 5 |
11 |
18 |
1 |
0 4 |
Total |
261 (73.3%) |
4 (1.1%) |
91 (25.6%) |
To determine on which chromosome of the standard karyotype Gld resides, we crossed a structural heterozygote from the above described F
2 with the testerline WL1255 possessing a genotype A/A 1221/1221. A heterozygote 1221/1123 with fully fertile pollen, i.e. a structural homozygote for the normal karyotype without the additional chromosome, was chosen from the F1. It gave rise to 211 F2 plants, of which only four had phenotype gld, all being cri (Table 4). The gld mutation assorted independently of the locus His(2-6) but showed linkage to cri, although the distance of 0.6 % cross-over obtained is not reliable due to a strongly biased monogenic segregation for both cri and gld. Table 4. Segregation of phenotypes in progeny of the plant with normal karyotype (1123/1221) taken from the cross of WL1255 (1221, Cri, Gld) with the structural heterozygote (2121/1123, Cri/cri, Gld/gld)1221 |
1221+1123 |
1123 |
|
| Cri Gld | 51 |
102 |
46 |
| cri gld | 1 |
2 |
1 |
| cri Gld | 3 |
3 |
4 |
Recombination fractions: cri - His(2-6)= 49.0 +/- 0.4, JSCS = 1.3; gld - His(2-6)=
47.5 +/- 4.2, JSCS–joint segregation chi-square = 0.003; cri - gld= 7.0 +/- 1.8, JSCS = 57.9, P <
0.001.
All linkages are significant at the 0.1% level
The evidence indicates that Gld is located on linkage group V distal to cri and possibly very close to it.
1. Berdnikov, V.A., Gorel, F.L. and Temnykh, S.V.
1993. Pisum Genetics 25: 18-20.
2. Lamm, R.L. 1986. PNL 18:34-37.
3. Pellew, C. 1940. J. Genet. 39:363-390.
4. Weeden, N.F., Ellis, T.H.N., Timmerman-Vaughan, G.M., Swecicki, W.K., Rozov, S.M. and
Berdnikov, V.A. 1998. Pisum Genetics 30: 1-4.